Inside EU Health: UK Tobacco and Vapes Bill; GLP-1 pill for type-2 diabetes in adolescents; maternity and paternity leave across the EU
UK Tobacco and Vapes Bill clears final hurdle; Novo Nordisk announces positive results for GLP-1s for teenagers with type 2 diabetes; Maternity and paternity leave across the EU-27

UK Tobacco and Vapes Bill clears final hurdle
A bill that will prohibit the sale of tobacco, vapes and other products, to people born on or after 1 January 2009, and that will control the advertising and promotion of tobacco, vapes and other products; as well as creating smoke-free, vape-free and heated tobacco-free places, is awaiting royal assent.
This means that as of 1 January 2027, anyone who is 18 or younger will never be able to buy cigarettes in the UK.
“Through the Tobacco and Vapes Bill we are taking bold action to create a smoke-free generation and protect future ones from the harms of tobacco,” said Public Health Minister Sharon Hodgson. “Alongside this, record investment in stop smoking services is supporting current smokers to quit.” The UK Secretary of State for Health, Wes Streeting, described it as “an historic moment”.
“We know there is a lot of cynicism about politics and whether governments can make a difference,” said Sarah Woolnough, CEO of health think tank The King’s Fund. “It is rare that there is the opportunity for a government to introduce a world-leading policy, that doesn’t cost a huge amount, and that will be remembered for generations as a moment when the dial was decisively shifted towards preventing disease and lives were saved.”
Initially proposed by the Conservative government under Rishi Sunak in 2024, the most vocal opposition comes from the Reform Party, led by Brexit supporting former MEP Nigel Farage, who promises to overturn the law should Reform come into power.

The EU’s beating cancer plan is also targeting a ‘Tobacco Free Generation’ by 2040, where less than 5% of the population uses tobacco by 2040, but a proposed Tobacco Taxation Directive is facing strong headwinds and the Commission is yet to present its revised Tobacco Products Directive and Tobacco Advertising Directive, expected later this year. A similar age ban is almost unthinkable at an EU-level, but many will be looking on to see if it is successful.

Novo Nordisk announces positive results for GLP-1s for teenagers with type 2 diabetes
Novo Nordisk has announced positive results from a phase 3 trial evaluating oral semaglutide for type 2 diabetes in children and adolescents aged 10–17 years. The trial showed a well-tolerated safety profile. Oral semaglutide is approved in the EU as Rybelsus® and is currently available to adults.
“Over the past two decades, the prevalence of type 2 diabetes among children and adolescents has increased substantially, yet treatment options for this population remain limited, underscoring a significant unmet need,” said Novo’s Chief Scientific Officer, Martin Holst Lange.
“These results from the PIONEER TEENS trial confirm that oral semaglutide is an effective treatment option for children and adolescents with type 2 diabetes who require glycaemic control beyond that provided by the current standard of care.”
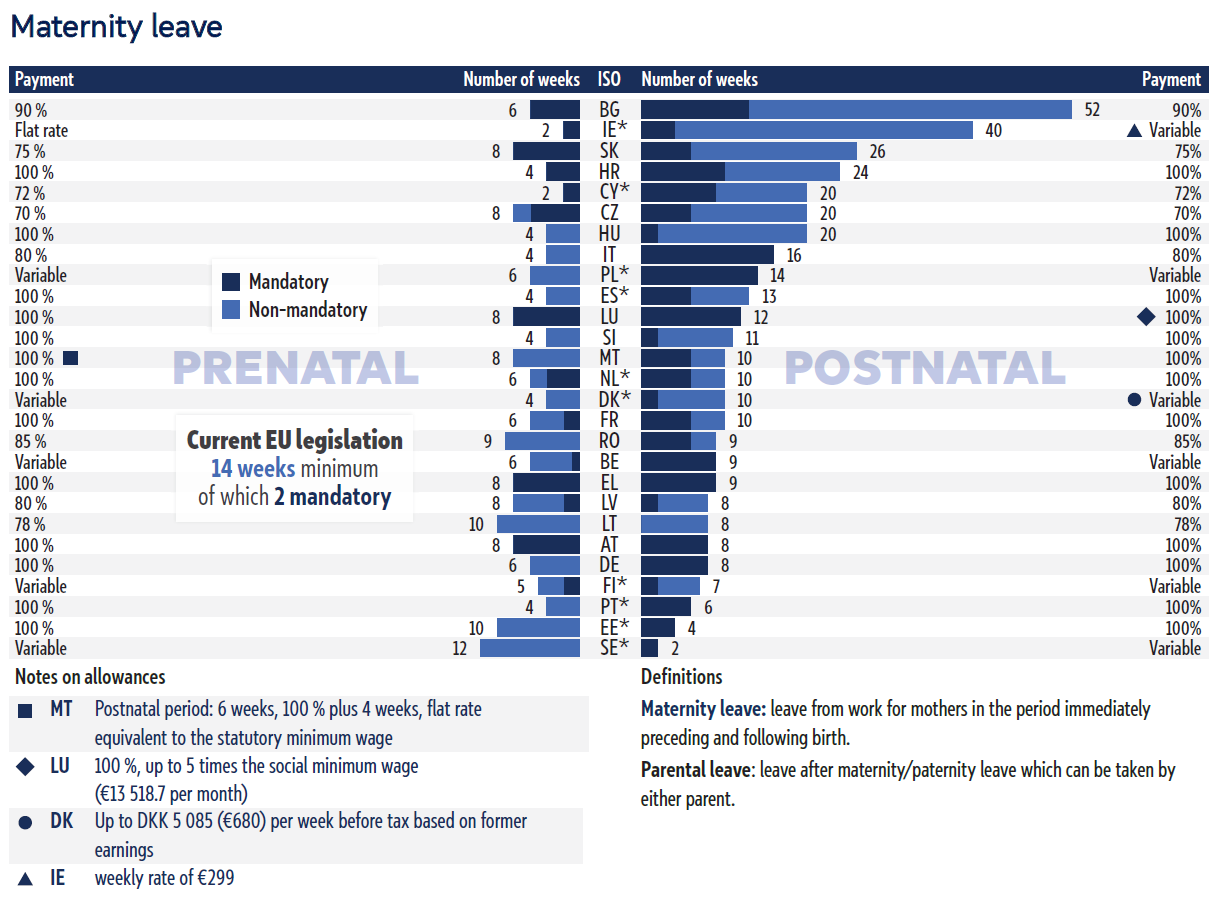
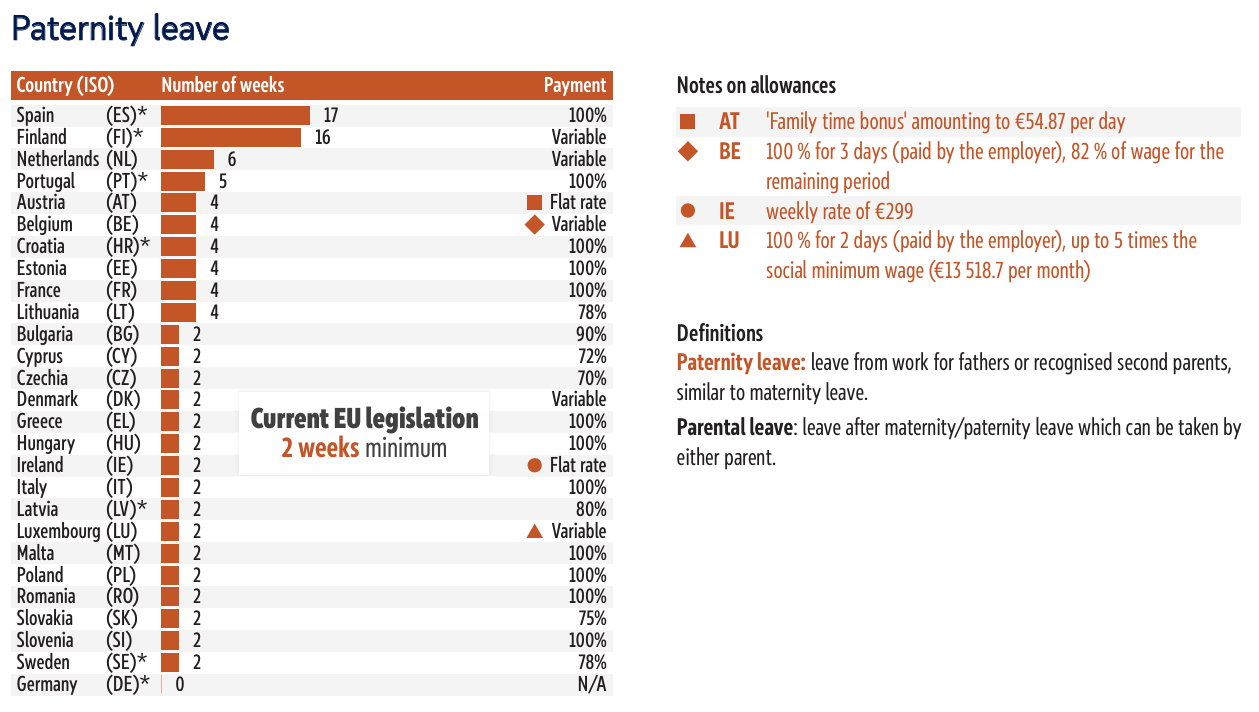
Maternity and paternity leave across the EU-27
The European Parliament Research Service has produced a report comparing maternity and paternity leave across the EU’s 27 Member States, highlighting the wide differences in duration, pay, and mandatory provisions.
While EU directives set minimum standards, 14 weeks of maternity leave under the Pregnant Workers Directive and two weeks of paternity leave under the Work-Life Balance Directive, many countries exceed them in varied ways.