Inside EU Health: Chemicals Omnibus; EU Pharma exports hit record high; EHDS
Cancer leagues urge MEPs to reject weaker cosmetic safety rules; Ireland leads EU pharmaceutical exports as trade surplus hits record high; European digital identity wallets and cross-border health data infrastructure

Cancer leagues urge MEPs to reject weaker cosmetic safety rules
The Association of European Cancer Leagues (ECL) has urged MEPs to reject proposed changes to EU cosmetics rules, warning they could weaken protections against cancer-causing substances.
In a letter ahead of key committee votes, ECL cautioned that the proposed ‘Chemicals Omnibus’ risks increasing exposure to harmful chemicals in everyday products and undermines commitments made in Europe’s Beating Cancer Plan.
ECL’s Executive Director Dr Wolfgang Fecke said lowering safeguards would “increase the risk of cumulative exposure to carcinogens” and marks “a significant step backward” for public health. The group called on lawmakers to maintain the EU’s precautionary approach, stressing that “protecting citizens from avoidable exposure” must remain “a non-negotiable priority” as Parliament considers amendments to the regulation.

Ireland leads EU pharmaceutical exports as trade surplus hits record high
New figures released by Eurostat show that the EU’s trade in medicinal and pharmaceutical products reached unprecedented levels in 2025, with Ireland emerging as the dominant exporter.
The EU recorded a trade surplus of €220.5 billion in medicinal and pharmaceutical products, driven by strong export growth and rising global demand. Total exports climbed to €366.2 billion, marking a 16.0% increase compared with 2024, while imports rose even faster by 21.0%, reaching €145.7 billion.
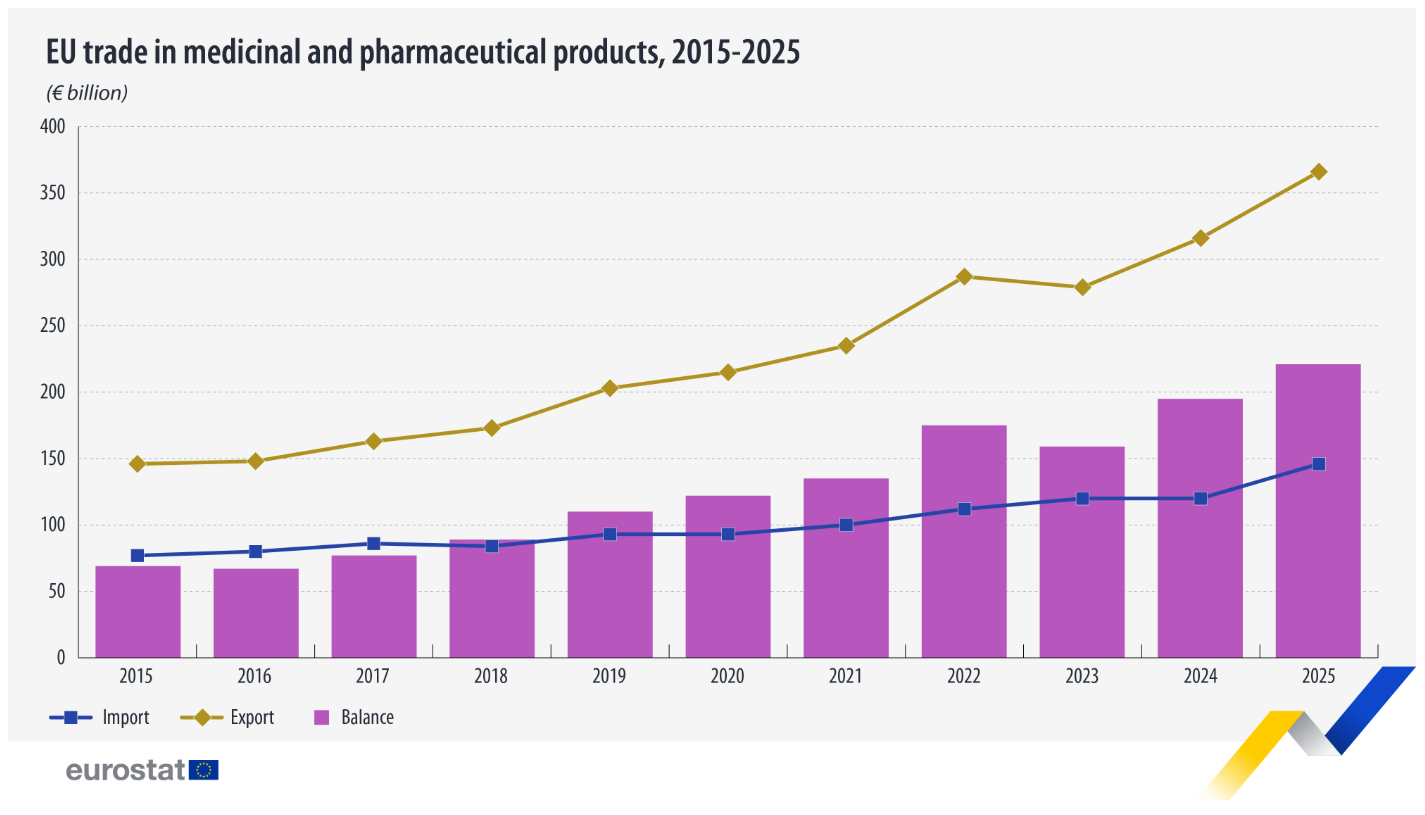
Ireland solidified its position as the EU’s leading exporter of pharmaceuticals to countries outside the EU, with exports valued at €93.8 billion. It was followed by Germany (€67.9 billion) and Belgium (€38.5 billion), highlighting the concentration of high-value pharmaceutical production in a handful of member states.
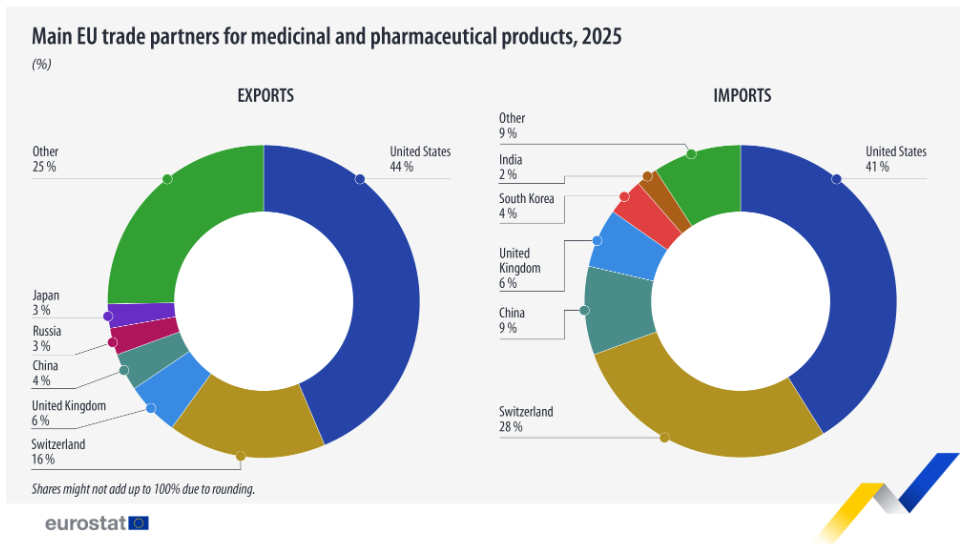
On the import side, Italy ranked as the EU’s largest buyer of medicinal and pharmaceutical products from outside the EU.
The United States continued to dominate as the EU’s primary external trading partner for pharmaceuticals, accounting for 43.8% of all EU exports. Followed by Switzerland (€59.7 billion, 16.3%), and the United Kingdom (€20.6 billion, 5.6%). On the import side, the United States again led as the EU’s largest supplier, providing €60.1 billion worth of products, or 41.2% of total imports.
European digital identity wallets and cross-border health data infrastructure
Following last week’s adoption of the European Health Data Space (EHDS) board, implementation continues apace, with the launch of two new public consultations on draft implementing acts, critical to the working of EHDS.
The first focuses on identity management. Accurate patient matching is essential when sharing data such as patient summaries. Under the proposal, ‘c will be used to streamline this process, enabling patients to securely provide the necessary details to link their medical records across countries. This would allow healthcare professionals abroad to quickly access relevant information when treating traveling patients.
The second concerns MyHealth@EU, the infrastructure that will underpin cross-border data exchange within the EHDS. It will ensure that patient summaries, ePrescriptions, and dispensation data are transmitted securely and reach the appropriate healthcare providers. Over time, the system is expected to expand to include additional data types, such as medical imaging.
Both consultations are open for feedback until 7 May, offering stakeholders an opportunity to contribute to shaping Europe’s digital health framework.

