Inside EU Health: ESF+ for abortion access; pharma reforms could reduce costs by 20-25%; pharma 29% of exports to US; ECCE receives lukewarm welcome
Commission open ESF+ pathway for abortion access; EU’s Health Package is Europe’s answer to Trump’s drug pricing policy, says Várhelyi; medical and pharmaceutical products were the leading export category to US in 2025; ECCE idea receives lukewarm welcome

Commission open ESF+ pathway for abortion access
The European Commission has confirmed that member states can use the European Social Fund Plus (ESF+) to support access to safe and legal abortion care, following the ‘My Voice, My Choice’ citizens’ initiative backed by 1.2 million Europeans.
Equality Commissioner Hadja Lahbib called the decision “groundbreaking,” saying, “From now on, we are granting member states the possibility to use EU funding… to improve access to abortion.” She added that the initiative proved “democracy can deliver.”
MEP Abir Al-Sahlani (Renew, Sweden), rapporteur for Parliament’s response to the initiative, described the decision as a turning point: “For the first time, the Commission formally confirms that EU funding can support abortion access. That is historic.”
Activists welcomed the move as historic. “Today is a victory for women in Europe,” said campaign coordinator Nika Kovač. For the first time, she noted, the Commission confirmed “unequivocally” that EU funds can help guarantee access to safe abortion care.

EU’s Health Package is Europe’s answer to Trump’s drug pricing policy, says Várhelyi
European Commissioner Oliver Várhelyi said the EU’s response to the Trump administration’s “most-favoured nation” drug pricing policy goes beyond tariffs and price controls. While tariffs are an EU competence, medicine pricing and healthcare systems remain national responsibilities, limiting Brussels’ direct role.
Instead, the Commission has focused on regulatory simplification in medical devices and biotech, aiming to cut administrative burdens without weakening safety or scientific standards. Várhelyi estimates the reforms could reduce business costs by 20-25 percent, potentially offsetting tariff pressure.
The broader strategy is to secure innovation, clinical trials, and manufacturing in Europe through targeted biotech incentives and extended protections for innovative products, while preserving universal access to top therapies.

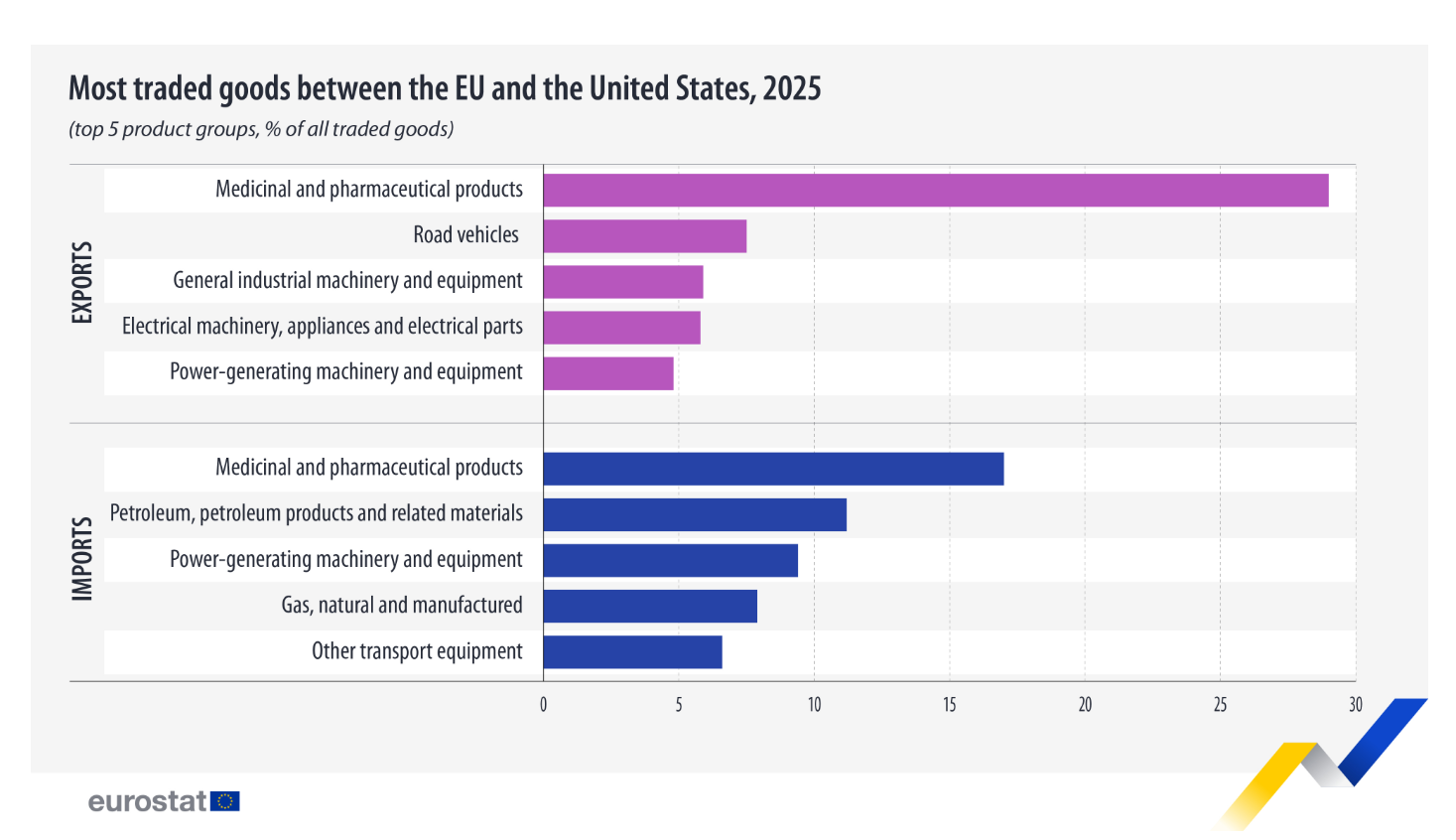
Medical and pharmaceutical products were the leading export category in 2025
Eurostat reports that in 2025, the European Union exported €554.0 billion worth of goods to the United States and imported €354.4 billion, resulting in a trade surplus of €199.6 billion. Compared with 2024, exports increased by 3.4%, while imports rose by 4.8%.
The five largest export groups accounted for more than half (53.0%) of total exports to the United States. Medicinal and pharmaceutical products (29.0%), road vehicles (7.5%), general industrial machinery and equipment (5.9%), electrical machinery, appliances and parts (5.8%) and power-generating machinery and equipment (4.8%)
ECCE receives lukewarm welcome
The idea of establishing a ‘European Centre of Clinical Excellence for Pharmaceuticals' seems to have received a lukewarm welcome.
Speaking after today’s Informal Health Council, Cypriot Health Minister Neophytos Charalambides said: “To be honest, the ministers expressed concerns today, as far as the finances, and whether this would interact in any way with the Health Technology Assessments work and other authorities, EMA etcetera. So we'll take all these concerns into consideration, and our intention is to go through the conclusions and move the discussion of this issue to the next level.”
In yesterday’s newsletter, we outlined the Cypriot proposal for the creation of a ‘European Centre of Clinical Excellence for Pharmaceuticals' (ECCE) intended to strengthen cooperation on clinical evaluation and the use of medicines across the European Union.
The Cypriots said the ECCE would be a “science-driven hub focused on clinical excellence” and would not replace existing European or national bodies, nor interfere with member states’ competences in pricing and reimbursement.


