Inside EU Health: AI; MDR simplification; European patents; Ireland supports ‘right to be forgotten’ law
AI law changes risk weakening patient safety in healthcare; EU extends ‘well-established technologies’ list under MDR; medical technology and pharmaceuticals are among top ten European patent fields; Ireland to make ‘right to be forgotten’ law for cancer survivors

AI law changes risk weakening patient safety in healthcare
The European Parliament is set to vote this week on amendments to the Artificial Intelligence (AI) Act that could significantly reshape how AI is regulated in European healthcare. While presented as a simplification, the proposed changes risk creating a dangerous regulatory gap, according to Health Action International (HAI).
HAI argues that AI medical devices must remain within the Act’s high-risk category and is urging MEPs to reject the proposals and maintain strong protections for patient safety and fundamental rights.
Without the safeguards of this categorisation, HAI say that clinicians may lack clear guidance, and healthcare providers may fail to implement necessary protections, raising the risk of misuse and patient harm. They are also concerned by biased training data, limited transparency, and unequal outcomes across patient groups.
Maintaining robust oversight, they argue, is essential to ensure AI improves healthcare without compromising safety or fairness.
EU extends ‘well-established technologies’ list under MDR
As part of its promise to simplify the bureaucratic burden on medical devices, the EU has published its delegated act expanding the list of ‘well-established technologies’ in the Medical Devices Regulation (MDR).
The list was extended following extensive consultation in 2025 with competent authorities and in the Medical Device Coordination Group, it builds on experience gained through the implementation of the MDR. The simplified procedure will reduce the legal and administrative burden for a number of implantable devices, such as sutures, cannulas, feeding tubes and much else.
Well-established technologies are medical devices that are considered simple, stable in design, and supported by a long history of safe clinical use. The extended list should not affect the high standards of safety and performance.
Medical technology and pharmaceuticals are among top ten European patent fields
The latest European Patent Office (EPO) Technology Dashboard 2025 shows an increase in overall patent applications, surpassing the 200,000 mark for the first time. Over a third of the applications were from EU27 states.
“The record volume of patent applications underlines Europe’s innovative capacity and its appeal as a global technology market,” said EPO President António Campinos. He said that the Technology Dashboard 2025 tracks progress and gaps across sectors and should help policymakers in Europe identify priority areas and target actions and investments to strengthen sovereignty and competitiveness.
European innovators led EPO applications in eight of the ten top technology fields, including medical technology and pharmaceuticals. Nevertheless, the number of pharmaceutical patents went down by 6.3% and those for medical technology increased by only 1.3%.
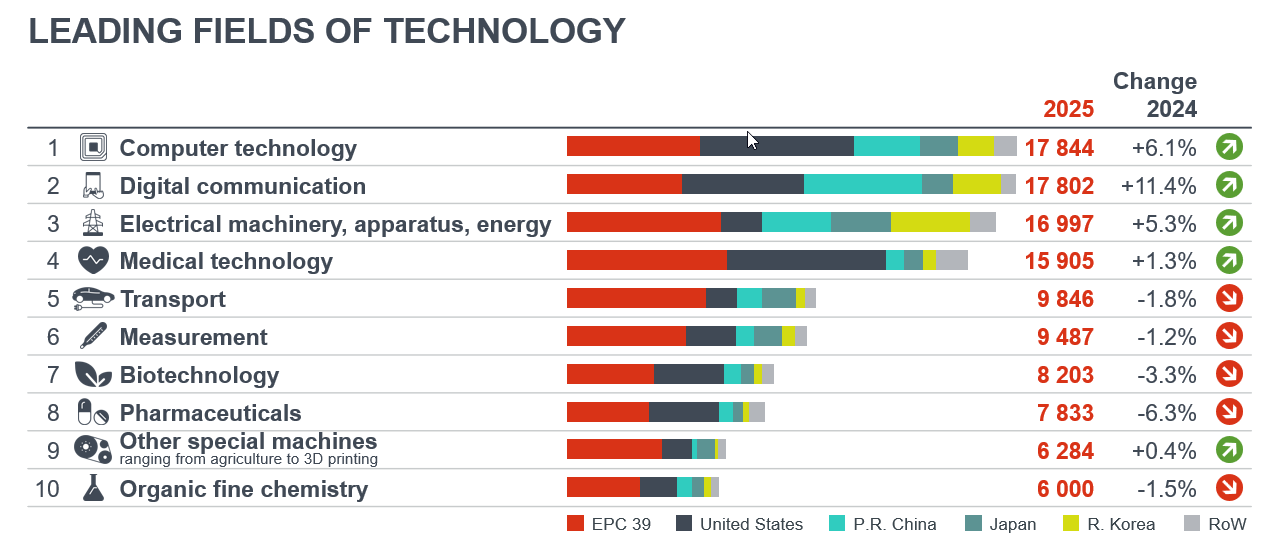
Europe maintains a strong position
Within Europe, growth was driven maily by mid-sized innovation economies, including Finland (+44%), Denmark (+5.2%), Austria (+5.0%), and Spain (+2.9%). Filings from some of Europe’s largest patenting countries declined slightly, including Germany and France.
Unitary patents
Unitary Patents, which make it possible to receive patent protection in up to 18 EU states by submitting a single request to the EPO, is up to 28/7% of all patents and 40.7% for patent owners from EU countries. Campinos said that while the Unitary Patent is already removing barriers and accelerating the transition to a more integrated European innovation market, continued focus was needed; he highlighted health as one sector that could do more.
Ireland to make ‘right to be forgotten’ law for cancer survivors
The Irish Government has taken a significant step toward introducing a legally enforceable ‘right to be forgotten’. The legislation aims to ensure that cancer survivors can access mortgage protection insurance on fair and equitable terms, replacing the current voluntary framework in place since December 2023.
The proposal will reduce the remission period required before a cancer survivor can be considered for mortgage protection insurance from seven years to five years. In addition, the threshold for mortgage protection cover will increase from €500,000 to €650,000, expanding access for homebuyers.
“A cancer diagnosis should never define somebody’s financial future or inhibit them from owning their own home. For too long, this sadly has been the case,” said Irish Deputy Prime Minister, Simon Harris.
In February, the European Commission announced that it would present guidance to financial undertakings on offering fair access to financial services later this year.


