Inside EU Health: CMA update; cardio diseases responsible for one-third of EU deaths; ECDC to develop RSV guidance
Critical Medicines Act slowly edging forward; Circulatory diseases are responsible for one-third of deaths in the EU in 2023; ECDC to present guidance on RSV vaccines for adults by 2027; Roche scales AI; AstraZeneca gastric cancer drug approved; Servier single-pill formulation to improve adherence

Critical Medicines Act slowly edging forward
As expected, yesterday’s Critical Medicines Act trilogue discussed the scope of the proposal. The Parliament and Council remain divided, with the Parliament trying to extend the scope to “medicines of common interest” and the Council resisting a definition that goes beyond a critical medicines list. Further work will be undertaken at a technical level.
Regarding recognition of strategic projects (Article 6), some convergence was reached on the timeline proposed by the Parliament that each EU state designate an authority within a three-month period following the regulation coming into force, some progress was made but technical talks continue.
The next trilogue will take place on 11 May, with negotiations seemingly still at an early stage, it might be difficult to reach an agreement before the end of the Cypriot Presidency in June.
Circulatory diseases responsible for one-third of deaths in the EU in 2023
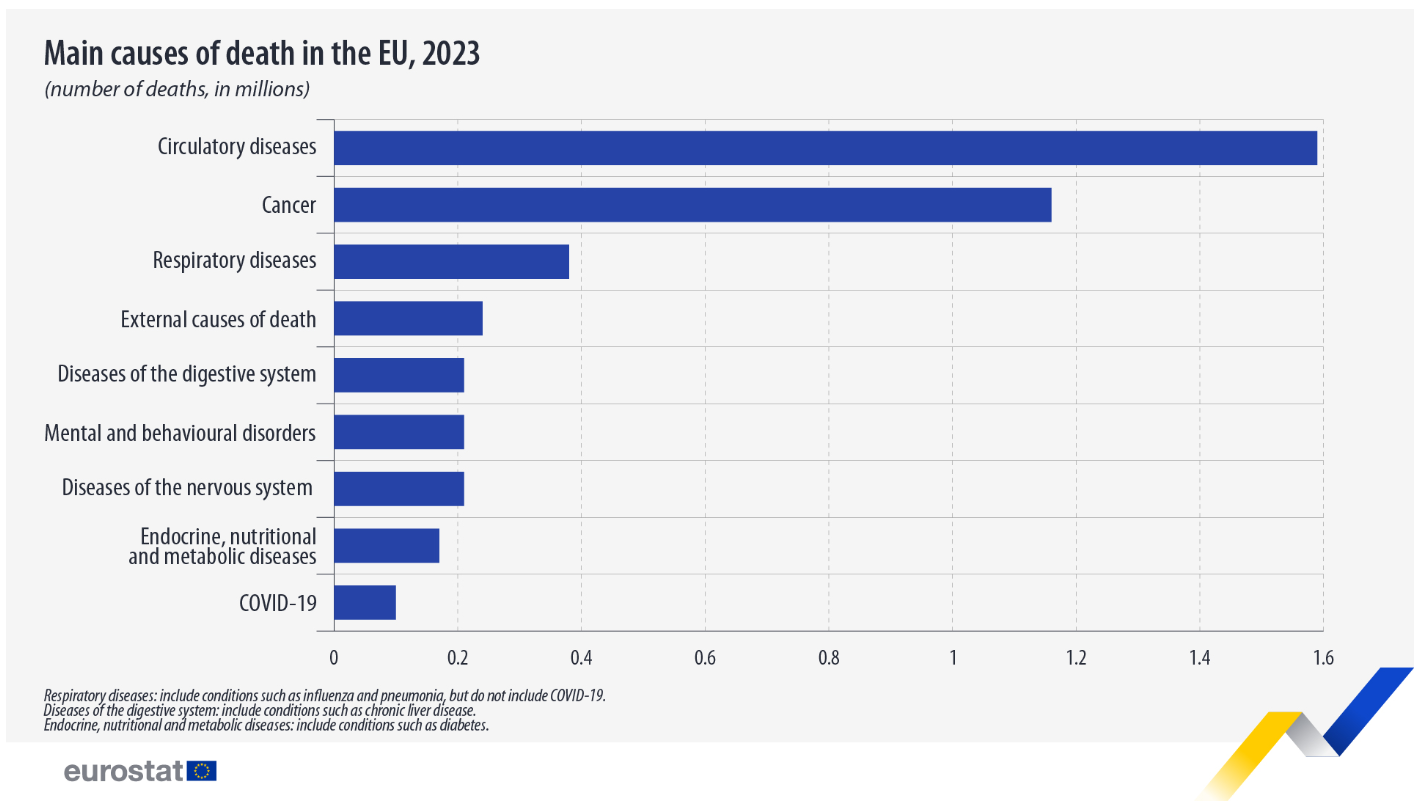
Circulatory diseases accounted for one-third of deaths in the EU in 2023. In total, 4.84 million people died in the European Union that year. Circulatory diseases are responsible for 1.59 million deaths (32.8%). Circulatory diseases include conditions such as high blood pressure, heart disease and diseases affecting the veins and arteries.
Cancer was the second most common cause of death, accounting for 1.16 million deaths (23.9%), followed by respiratory diseases - excluding COVID-19 - with 0.38 million deaths (7.8%).
Other major causes of death included external causes of morbidity and mortality (5.0%), diseases of the digestive system (4.4%), mental and behavioural disorders (4.3%), diseases of the nervous system (4.3%), endocrine, nutritional and metabolic diseases (3.5%) and COVID-19 (2.1%).
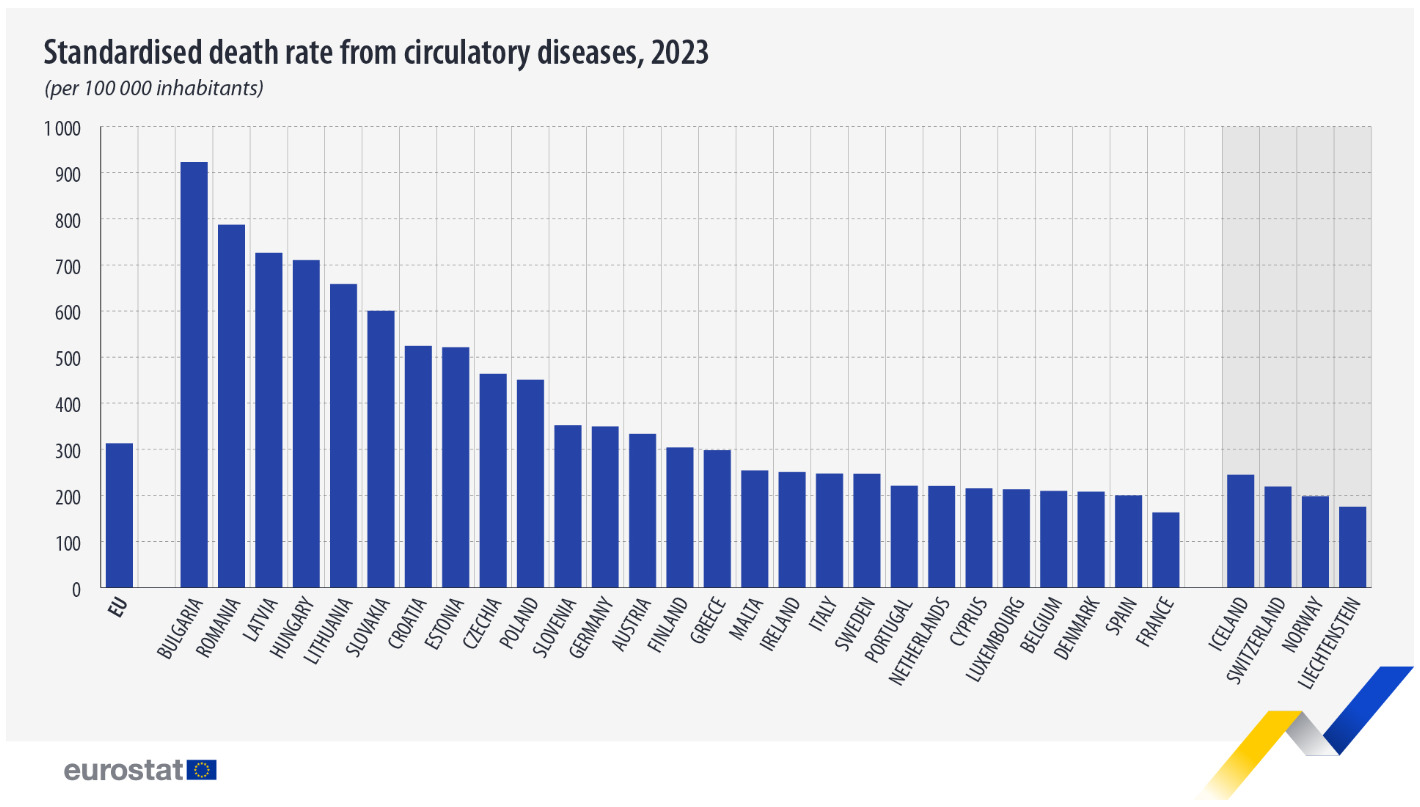
The figures across the EU show and East/West divide the highest death rates from circulatory diseases were recorded in Bulgaria (923 deaths per 100,000 people), Romania (787) and Latvia (726). In contrast, the lowest rates were observed in France (163), Spain (200) and Denmark (208).
ECDC to present guidance on RSV vaccines for adults by 2027
The European Centre for Disease Prevention and Control (ECDC) has launched a call for experts to present guidance on the use of respiratory syncytial virus (RSV) vaccines to prevent disease in adults, with publication planned for mid-2027.
The guidance will support EU and European Economic Area countries as they consider vaccination strategies against RSV, a respiratory virus that causes around 160,000 hospitalisations among older adults in Europe each year.
The document will propose non-binding recommendations that countries can adapt to their national public health contexts. The guidance will review vaccine safety and effectiveness, including co-administration with other vaccines, and the duration of protection and possible need for re-vaccination.
Three RSV vaccines are currently authorised in Europe: Arexvy (GSK), Abrysvo(Pfizer), and mResvia (Moderna). All are approved for adults aged 60 and over. Abrysvo is also authorised for adults aged 18 and above, while mResvia can be used in adults over 18 with risk factors for severe RSV disease.
Roche scales AI with NVIDIA
Roche has announced an expansion of its global AI infrastructure, deploying a large-scale AI factory powered by NVIDIA high-performance GPUs on premises across the United States and Europe.
“Our AI factory combines world-class computing power with Roche’s scientific expertise to embed AI across the entire value chain - from discovery to development, manufacturing and commercialisation - transforming how we deliver the next generation of medicines and diagnostics solutions,” said Roche’s Chief Digital and Technology Officer, Wafaa Mamilli.
AstraZeneca welcomes EU approval of gastric cancer immunotherapy drug
AstraZeneca’s Imfinzi (durvalumab) in combination with chemotherapy has been approved in the EU for the treatment of adult patients with resectable, early-stage and locally advanced gastric and gastroesophageal junction cancers.
Gastric cancer is the fifth leading cause of cancer death globally, with nearly one million people diagnosed each year.
In a Phase III clinical trial, nearly 70% of patients were still alive three years after treatment with the durvalumab-based perioperative regimen.
“This EU approval brings patients the first immunotherapy regimen to extend survival in this early setting and is poised to become the new standard of care,” said the trial's principal investigator, Director of the Vall d’Hebron Institute of Oncology (VHIO) in Barcelona, Spain Josep Tabernero, MD, PhD.
“For patients with early gastric and gastroesophageal cancers, this immunotherapy-based regimen delivers a durable survival benefit,” said Executive Vice President, Oncology Haematology Business Unit, AstraZeneca, Dave Fredrickson.
Servier develops a single pill to improve adherence
Servier is developing new drug formulations that reduce the number of pills a patient needs to take by combining active ingredients into a single gelcap. The single-pill combinations also enable new dosages and more precise targeting of patients’ needs. Improving adherence slows the progression of chronic conditions and make it easier for patients to adhere to regimens.
